
2
1
Functional Group
Characteristics and Roles
INTRODUCTION
This chapter is written with the assumption that the reader has a basic knowledge of
organic chemistry and is at least familiar with the terminology used to describe the
parts of an organic molecule. The goals of this chapter are to define the term functional
group, review the major chemical properties or characteristics inherent to any given
functional group, relate these chemical properties or characteristics to the discipline
of medicinal chemistry, and to provide some initial examples of how these properties
or characteristics are important for drug action.
Subsequent chapters provide detailed information with regard to the ionization
of acidic and basic functional groups, the roles of water and lipid soluble functional
groups, the types of chemical interactions possible between functional groups and their
biological targets, the specific routes of metabolism associated with specific functional
groups, and how functional groups can be altered to provide a therapeutic benefit.
LEARNING OBJECTIVES
After completing this chapter, students will be able to:
1. Identify the individual functional groups that comprise the structure of a given
drug molecule.
2. Explain the general purpose of functional groups and provide specific examples
of how functional groups affect drug activity.
3. Analyze the electronic, solubility, and steric effects that an individual functional
group can impart to a specific drug molecule.

2 Basic Concepts in Medicinal Chemistry
4. Explain how a specific functional group can serve different purposes on differ-
ent drug molecules and how the importance of a specific functional group can
vary among different drug molecules based on the influence of the adjacent
functional groups.
5. Explain how functional groups can affect therapeutic outcomes.
6. Identify the key chemical properties of the functional groups present in amino
acids and proteins.
WHAT IS A FUNCTIONAL GROUP?
Prior to answering this question, let us begin with two objects for which everyone is
familiar, an automobile and a refrigerator. Each of these machines consists of hundreds
of interrelated parts that are essential for specific functions. Some of these functions
are absolute requirements, while others are desired but not considered required. As
an example, let’s consider the wheels. This part of the automobile is essential to allow
it to move quickly and smoothly over hundreds and thousands of miles. In contrast,
the wheels found on most modern refrigerators are helpful in moving it a few feet for
cleaning purposes or perhaps to remodel a kitchen, however they are not considered
essential. A refrigerator without wheels is still fully functional and it is still possible
to move it, either using a dolly or by a couple of people pushing it. On the other
hand, an automobile without wheels is no longer functional. Table 2-1 outlines some
additional comparisons between these objects. The key point of this initial example
is to emphasize that these familiar objects have different uses and functions, contain
both similar and different parts, and that the relative need of a given specific part
varies depending upon the object. Some parts, such as a power source, are essential
for almost all machines, while others, such as a mirror or a horn, are not.
Table 2-1. A comparison of the specifi c requirements for selected parts of an
automobile and a refrigerator
Part Automobile Refrigerator
Power source Essential. Requires gasoline, electricity or a hybrid. Essential. Requires electricity.
Doors Essential. Required to enter and exit the vehicle. Essential. Required to access food and keep
items at proper temperature.
Windows Essential. Required to see the road, pedestrians,
traffi c signs, etc. Power windows are somewhat
standard, but really not required.
Not needed.
Thermostat and
coolant system
Essential. Required to ensure the engine does not
overheat.
Essential. Required to keep food and drinks at
desired temperature.
Lights Essential. Required for nighttime driving and
signaling stops and turns.
Highly desired but not essential.

Chapter 2: Functional Group Characteristics and Roles 3
Similar to automobiles, refrigerators, and other machines, drug molecules consist
of various components known as functional groups. This is exemplified by fluoxetine,
an antidepressant that selectivity blocks the reuptake of serotonin. This drug is com-
prised of seven parts or seven specific functional groups.
Ether ox
yg
en
Alk
y
l chain
Secondar
y
amine
Meth
y
l
g
rou
p
Aromatic rin
g
Aromatic rin
g
Trifluorometh
y
l
g
rou
p
Defi nitions
Pharmacokinetic effects have been generally defi ned as those that
explain what the body does to the drug, whereas pharmacodynamics
effects have been generally defi ned as those that explain what the drug
does to the body. More specifi cally, pharmacokinetic effects include
the absorption, distribution, metabolism, and elimination (ADME) of a
drug molecule, while pharmacodynamic effects include the intensity,
duration, and mechanism of action of that same drug molecule. Intrinsic
within a pharmacodynamic effect is the ability of the drug molecule to
interact with its biological target. Biological targets can be organized
into four general categories (receptors, enzymes, nucleic acids, and
excitable membranes/other biopolymers) and are typically composed
of proteins or nucleic acids.
From a medicinal chemistry perspective, functional groups provide specific proper-
ties and behaviors that allow drug molecules to exert their desired pharmacodynamic
and pharmacokinetic effects. For a given drug molecule they play a significant role
in the:
• overall water/lipid solubility
• route of administration
• ability to interact with specific biological targets
• mechanism of action
• route of metabolism and elimination

4 Basic Concepts in Medicinal Chemistry
• duration of action
• suitability for a specific therapeutic situation
• tendency to cause adverse effects or drug interactions
An Exclusive Interview with Some Functional Groups
It is a rare occasion when a functional group will grant an interview;
however, the following fi ve functional groups have agreed to talk with
Tongue-In-Cheek Productions. Functional groups A–C reside within the
structure of enalaprilat, an angiotensin converting enzyme inhibitor that
is used to treat hypertension and other cardiovascular diseases; while
functional groups D and E residue within the structure of terbutaline,
a selective
2
agonist that is indicated for the treatment of asthma and
chronic obstructive pulmonary disease (COPD). Here are their responses
to the question, “What’s your job?”
Functional Group A: “I provide the initial ionic bond that allows this
drug to interact with its target enzyme.”
Functional Group B: “I interact with a zinc atom involved in normal
substrate catalysis.”
Functional Group C: “I interact with a hydrophobic site and greatly
enhance binding.”
Functional Group D: “I moved one carbon away from the other OH
group in order to decrease the metabolism of our molecule. I also
help to provide selective
2
action.”
Functional Group E: “I provide selectivity for receptors located in the
pulmonary system that helps to decrease unwanted side effects.”
Functional
Grou
p
A
Functional
Grou
p
B
Functional
Grou
p
C
Functional
Grou
p
D
Functional
Grou
p
E
Enala
p
rilat Terbutaline
Hopefully, these testimonials will help you understand why the term
functional groups is used to describe the pieces or parts of a drug
molecule. The key point here is that each individual group within a
drug molecule can serve to provide one or more specifi c roles, tasks,
or functions. As evidenced by functional groups A and B, the same
functional group—a carboxylic acid in this case—can serve different roles
depending upon its location within the structure of the drug molecule.

Chapter 2: Functional Group Characteristics and Roles 5
When examining drug molecules, there are three overriding concepts that you
should always consider. First, every atom within the structure of a drug molecule is part
of a specific functional group. This was shown above with fluoxetine. An additional
example of this concept is shown below using the nonsteroidal anti-inflammatory
agent, indomethacin.
Methox
y
g
rou
p
Indole rin
g
Aromatic
(p
hen
y
l
)
rin
g
Chlorine atom
(
a halo
g
en
)
Meth
y
lene
g
rou
p
Meth
y
l
g
rou
p
Carbox
y
lic acid
Carbon
y
l
g
rou
p
(p
art of an amide with
the indole nitro
g
en atom
)
Indomethacin
Second, within any given drug molecule or class of drug molecules, some functional
groups will be more important than others. This will vary among drug molecules
and drug classes. As an example, consider the presence or absence of a simple methyl
group. As shown in Figure 2-1, bethanechol and simvastatin have an additional methyl
group as compared to acetylcholine and lovastatin, respectively. The methyl group in
bethanchol offers several advantages for bethanechol over acetylcholine as acetylcho-
line non-selectively interacts with both muscarinic and nicotinic receptors, is rapidly
metabolized by acetylcholinesterase, and is not orally active. The additional methyl
group in bethanechol allows it to selectively interact with muscarinic receptors and
prevents its degradation by acetylcholinesterase, thus allowing it to be administered
as an oral tablet. In comparison, while the additional methyl group in simvastatin
does enhance its overall activity as compared to lovastatin, both drugs can produce
very similar actions in lowering plasma cholesterol levels. Other pharmacological and
pharmaceutical properties of these two drugs are essentially identical. Thus, while the
methyl group in bethanechol is essential for its activity, duration and route of admin-
istration, the methyl group in simvastatin, in comparison, is of much less importance.

6 Basic Concepts in Medicinal Chemistry
Acet
y
lcholine: R = H
Bethanecol: R = CH
3
Lovastatin: R = H
Simvastatin: R = CH
3
Figure 2-1. A comparison of the relative importance of an extra methyl group.
Third, it is possible to alter functional groups to enhance activity, increase absorp-
tion, decrease adverse effects, or provide other therapeutic benefits. An example of
this can be seen with acyclovir (Figure 2-2). Acyclovir demonstrates very poor oral
absorption. Alteration of the highlighted hydroxyl group to a valine ester provides a
significant increase in oral absorption. An additional example is seen with terbutaline
in Tongue-In-Cheek’s “An Exclusive Interview with Some Functional Groups.” The
movement of an aromatic hydroxyl group (Functional Group D) from one carbon
atom to another led to the enhanced duration and increased selectivity observed with
terbutaline.
Valac
y
clovirAc
y
clovir
Figure 2-2. Acyclovir and valcyclovir.
CHEMICAL PROPERTIES OF FUNCTIONAL GROUPS
There are three major chemical properties that need to be considered for every func-
tional group. Each functional group has an electronic effect, a solubility effect, and a
steric effect that needs to be considered when evaluating the overall pharmacodynamic
and pharmacokinetic properties of any given drug molecule. Prior to proceeding, there
are two key points to keep in mind. First of all, the addition of a single functional group
to a given molecule will affect the overall electronics, solubility, and steric dimensions
of that molecule. It is impossible for a functional group to alter only one of these
Chapter 2: Functional Group Characteristics and Roles 7
properties. As an example, consider a drug molecule that contains an unsubstituted
phenylethyl group. The addition of a para hydroxyl group will influence the electron
density of the phenyl ring through its ability to interact with the aromatic electrons.
Phen
y
l
Grou
p
Eth
y
l Grou
p
Additionally, the ability of this hydroxyl group to form hydrogen bonds will in-
crease the water solubility of the compound. Finally, since the hydroxyl group is larger
than the original hydrogen atom, its addition changes the overall steric dimensions of
the compound. The specifics of all of these changes will be subsequently addressed;
however, the key point here is that a single functional group can affect the overall
electronic, solubility, and steric profile of a drug molecule. The second key point is that
the overall effect of a given functional group depends upon all of the other functional
groups surrounding it or attached to it. As will be discussed in the next section, the
presence or absence of adjacent functional groups can drastically affect the chemical
properties of a specific functional group.
Electronic Effects
The electronic effect of a functional group is measured by its ability to either donate
its electrons to adjacent atoms or functional groups or to pull or withdraw electrons
away from adjacent atoms or functional groups. There are two main components that
comprise the overall electronic effect of a functional group, its ability to participate in
resonance and its intrinsic inductive effects. Let us examine each of these components
separately.
Resonance occurs when electrons are shared among a group of atoms that have
adjacent double bonds and lone pairs of electrons. Since the electrons are being
equally shared, the overall structure is actually a hybrid of all of the possible resonance
structures. Examples of resonance structures can be seen in Figure 2-3. As seen with
the carboxylic acid, resonance structures can exist within a functional group. In this
example, the negative charge is being equally shared across the two oxygen atoms.
The ability to allow a positive or negative charge to be shared among multiple atoms
is extremely important since it enhances the acidity or basicity of specific functional
groups. This will be discussed in more detail in Chapter 3. Resonance structures can also
occur when a functional group donates or withdraws electrons from adjacent groups.
An aromatic hydroxyl group, also known as a phenol, can share its electrons with the
adjacent aromatic ring. In this case, the oxygen donates its electrons to the aromatic
ring. As illustrated by the resonance structures, the negative charge can be equally

8 Basic Concepts in Medicinal Chemistry
shared by the three aromatic carbon atoms either ortho or para to the phenolic group.
In contrast to a phenolic group, a nitrile group will withdraw or remove electrons from
the aromatic ring. In this case, the nitrile group acquires a negative charge, and the
aromatic ring acquires a positive charge. Similar to the phenolic group, the positive
charge can be equally shared among the aromatic carbons ortho or para to the nitrile.
Resonance structures
of a carboxylic acid
Resonance structures of an
aromatic hydroxyl group
Resonance structures of an
aromat ic nitrile
g
rou
p
-
-
-
ortho
ortho
p
ara
N
–
C
R
R
H
O
R
H
O
R
CN
–
R
NC
H
O
R
H
O
R
N
–
C
R
RO
O
–
O
O
–
R
+
+
+
+
+
+
Figure 2-3. Examples of resonance structures.
The intrinsic inductive character or nature of an atom or functional group depends
upon its overall electronegativity, a chemical property that defines the ability of an
atom or functional group to attract electrons towards itself and away from other atoms
or functional groups. The larger the electronegativity the greater ability of an atom
or functional group to attract electrons. The electronegativity values for atoms com-

Chapter 2: Functional Group Characteristics and Roles 9
monly seen in drug molecules are shown in Table 2-2. There are a few key points to
remember when looking at this table.
1. Fluorine, oxygen, chlorine, and nitrogen have the highest electronegativities,
respectively, among all of the atoms in the periodic table.
2. With the sole exception of fluorine, oxygen will inductively attract electrons
from all other atoms.
3. Oxygen, nitrogen, and all four halogens (i.e., F, Cl, Br, and I) will inductively
attract electrons from carbon.
4. Carbon will inductively attract electrons from hydrogen.
5. The above inductive effects (due to differences in electronegativity) will create
a partial charge separation between the atoms comprising a functional group.
This partial charge separation results in a dipole. Two examples of dipoles are
shown below. These dipoles are very important in enhancing water solubility
(discussed later in this chapter) and allow a drug molecule to interact with its
biological target (discussed in depth in Chapter 6).
Table 2-2. Electronegativity Values for Atoms Commonly Seen in Drug Molecules
Atom Electronegativity Value
F 3.98
O 3.44
Cl 3.16
N 3.04
Br 2.96
I 2.66
S 2.58
C 2.55
H 2.20
P 2.19
G
+
G
-
Arrows indicate the
direct of electron movement
(
i.e.
,
the di
p
ole created
b
y
the inductive effect
)
G
+
G
-
R
1
O
R
2
R
2
O
H
R
1
As previously mentioned, the presence or absence of adjacent functional groups can
affect the chemical properties of a given functional group. Let us revisit the phenyl-
ethyl group to see how an adjacent phenyl ring can affect the overall electronic effect

10 Basic Concepts in Medicinal Chemistry
of a hydroxyl group. Figure 2-4 shows the original phenylethyl group along with
two analogs, one that contains an aromatic hydroxyl group and one that contains an
aliphatic (i.e., non-aromatic) hydroxyl group. As discussed above and shown in Fig-
ure 2-3, the aromatic hydroxyl group of Analog A is involved in resonance with the
adjacent aromatic ring. This ability to form multiple resonance structures overrides
the inductive effect of the oxygen atom and allows this functional group to act as an
electron donating group. In contrast, the aliphatic hydroxyl group of Analog B cannot
participate in resonance. Thus, its electronic effect is solely the result of its inductive
effect. Since oxygen is more electronegative than carbon, an aliphatic hydroxyl group
acts as an electron withdrawing group.
Analog A Analog B
Phenyl
Group
Ethyl Group
Figure 2-4. Varying electronic effects of a hydroxyl group based upon adjacent functional
groups. The arrows represent the fl ow of electrons.
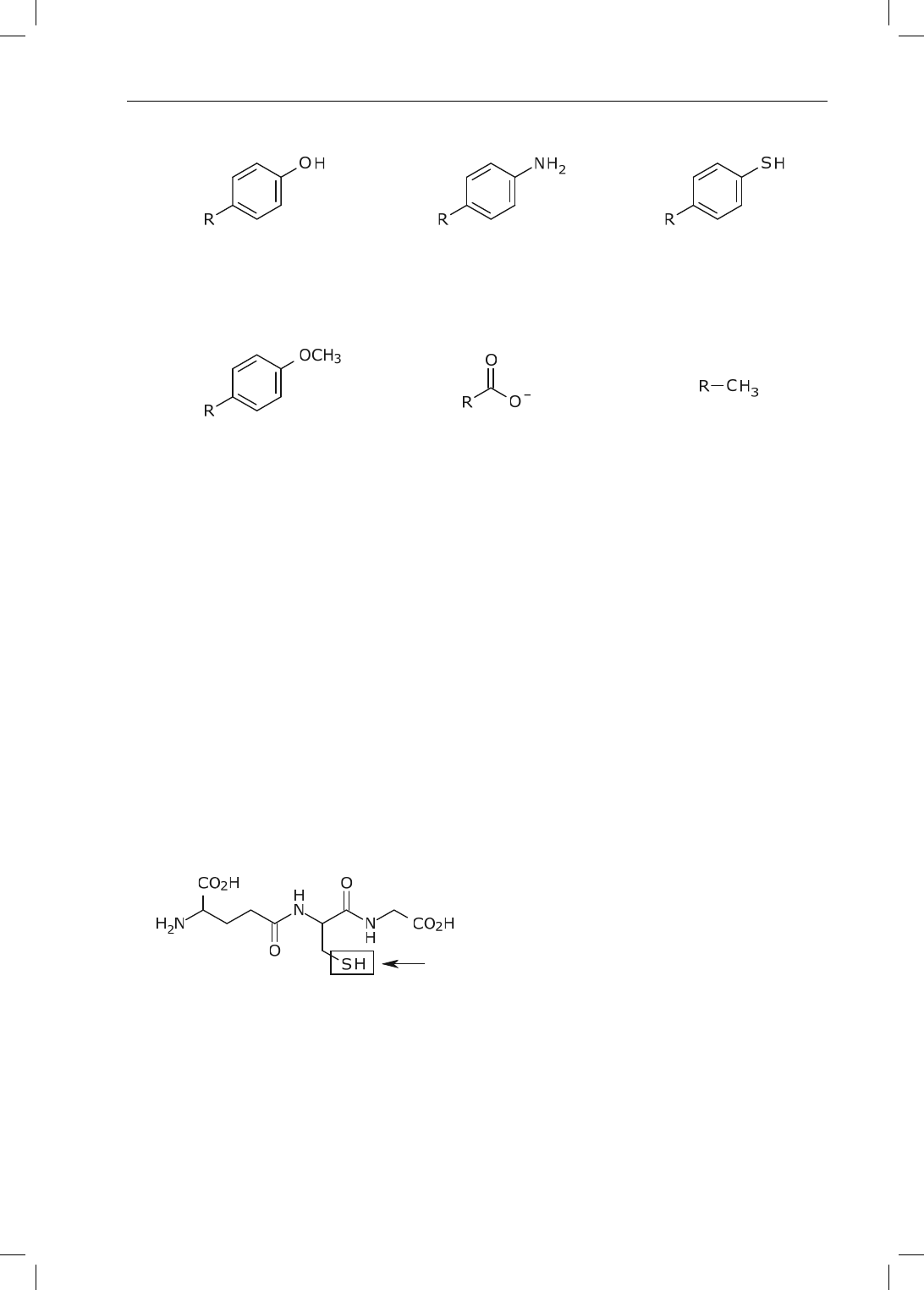
Electron Donating Functional Groups
The most commonly seen electron donating groups are shown in Figure 2-5. A few
key points need to be made here. First, negatively charged functional groups, such
as a carboxylic acid, can donate electrons through induction. This is also true for the
other acidic functional groups that will be discussed in Chapter 3. Second, functional
groups that contain a lone pair of electrons, such as a hydroxyl group, an aromatic
amine, an aromatic thiol, or a methoxy group, can donate electrons into a phenyl or
aromatic ring system as previously discussed. Finally, alkyl groups, such as a methyl
group or an ethyl group can serve as electron donating groups through induction.
Chapter 2: Functional Group Characteristics and Roles 11
Aromatic h
y
drox
y
l
(
i.e.
,
Phenol
)
Aromatic amine
(
Can be
p
rimar
y,
secondar
y,
or tertiar
y)
Aromatic thiol
Aromatic ether
(
Methox
y
g
rou
p
shown
)
Ionized acidic
g
rou
p
(
Carbox
y
lic acid shown
)
Alk
y
l
g
rou
p
(
Meth
y
l shown
)
Figure 2-5. Common electron donating groups.
Some of the electron donating groups mentioned previously can also serve as
nucleophilic functional groups. As the name implies, a nucleophilic group (i.e., a nu-
cleophile) is “nucleus loving” and thus attracted to the positive charge present in the
nucleus of an atom. Nucleophilic groups contain either a negative charge or a lone
pair of electrons that can be used to form a covalent bond with a biological target,
drug molecule or endogenous compound. Nucleophilic functional groups are involved
in the formation of hydrogen bonds, as well as with the irreversible bonds formed in
the mechanisms of alkylating agents, a select number of drug molecules, and a few
endogenous compounds (e.g., the sulfhydryl group found on glutathione). As will be
discussed in Chapter 8, this sulfhydryl group is very important in the inactivation of
highly reactive intermediates.
Glutathione
nucleo
p
hile
(
contains lone
p
airs of electrons
)
Electron Withdrawing Functional Groups
The most commonly seen electron withdrawing groups are shown in Figure 2-6.
Similar to electron donating groups, there are a few key points to remember. First,
halogens, a trifluoromethyl group, as well as positively charged functional groups,
such as an ionized amine, will pull or withdraw electrons through induction. Second,
when hydroxyl groups, sulfhydryl groups, and ether groups are not adjacent to either

12 Basic Concepts in Medicinal Chemistry
an aromatic ring or a double bond system, they act as electron withdrawing groups as
a result of their inductive effects. Finally, all of the other groups shown in Figure 2-6
can withdraw electrons through either resonance or induction. Adjacent functional
groups, as well as the presence or absence of direct attachment to an aromatic ring,
will determine the relative involvement of these two processes.
Nitro
g
rou
p
Halo
g
ens
(
X = F
,
Cl
,
Br
,
or I
)
Nitrile
g
rou
p
Trifluorometh
y
l
H
y
dox
y
lSulfh
y
dr
y
l or Thiol Ether
(
Methox
y
shown
)
Aldeh
y
de Amide Ester
Aromatic heteroc
y
cle
(
Isoxazole rin
g
shown
)
Sulfonamide
Ionized basic
g
rou
p
(
Primar
y
amine shown
)
R
N
O
R
O
NH
2
CNR
R
OCH
3
OCH
3
O
R
RCF
3
H
O
R
SHR
SNH
2
O
O
R
RNH
3
+
ROH
RNO
2
RX
Figure 2-6. Common electron withdrawing groups.
As opposed to electron donating groups that can serve as nucleophiles, some
electron withdrawing groups can also serve as electrophilic functional groups. As the
name implies, an electrophilic group (i.e., an electrophile) is “electron loving.” Thus,
examples of electrophilic functional groups are those that contain positive charges or a
good leaving group, such as a halogen or an ester. As alluded to previously, a nucleo-
philic functional group can attack an electrophilic functional group in order to form
a covalent bond. An example of this is shown with the nitrogen mustards. This class
of antineoplastic drugs forms an aziridinium intermediate as part of its mechanism.
This positively charged, three-membered ring is highly electrophilic and quickly forms
a covalent bond with a nucleophile.

Chapter 2: Functional Group Characteristics and Roles 13
Aziridinium Ion
(
Electro
p
hile
)
:
To close out this section, let us examine a specific situation where an electron with-
drawing group bestows a therapeutic benefit. Penicillin G and penicillin V are naturally
occurring penicillins; however, the oral bioavailability of penicillin V is superior to
that seen in penicillin G. As seen in Figure 2-7, the only structural difference between
penicillin G and penicillin V is the presence of an ether oxygen atom in penicillin V.
Before proceeding, let us evaluate this ether oxygen atom. As previously discussed, the
oxygen atom of an ether can be either electron donating (via resonance) or electron
withdrawing (via induction). In the case of penicillin V, it is both. The ether oxygen can
donate electrons to the adjacent aromatic ring via resonance and withdraw electrons
via induction from the adjacent methylene carbon and the other atoms attached to
this methylene unit. This results in an overall electron flow that withdraws electrons
from the right side of the drug molecule and donates them into the phenyl ring as
illustrated in Figure 2-7. It is this electron flow, specifically the electron withdrawing
effect, that allows penicillin V to have a better oral bioavailability than penicillin G.
Phenyl ring
Ether oxygen
Side chain
carbonyl
Penicillin G Penicillin V
E-Lactam ring
Elec ton donating
into phenyl ring
(resonance)
Elec tron withdrawing
into the ether oxygen atom
(induc tion)
Penicillin V
N
O
S
N
H
O
O
COOH
O
O
N
H
S
N
O
COOH
O
N
H
S
N
O
COOH
Figure 2-7. Electron fl ow of penicillin G and penicillin V.

14 Basic Concepts in Medicinal Chemistry
In the acidic environment of the stomach, penicillin G undergoes significant acid-
catalyzed degradation. The key mechanistic step involves the lone pair of electrons
present on the side chain carbonyl group. In an acidic environment, these electrons
attack the carbonyl carbon of the -lactam ring. The ultimate effect is destruction
of the -lactam ring in the stomach and inactivation of penicillin G. In contrast, the
presence of the ether oxygen, withdraws the lone pair of electrons from the side chain
carbonyl as discussed above. This decreases the availability of the lone pair of elec-
trons to attack the carbonyl carbon of the -lactam ring, thus allowing penicillin V
to be much more stable in the acid environment of the stomach. From a therapeutic
standpoint, both of these drugs have a similar spectrum of antibiotic activity; how-
ever, penicillin V can be used orally, while penicillin G must be administered either
intravenously (IV) or intramuscularly (IM).
::
::
Electon flow in an acidic
environment for Penicillin V
Electon flow in an acidic
environment for Penicillin G
O
O
N
H
S
N
O
COOH
O
N
H
S
N
O
COOH
Solubility Effects
The overall water and/or lipid solubility of a drug molecule affects its route(s) of
administration, distribution within the body, metabolism, duration of action, and
route(s) of elimination. This overall solubility is a composite sum of the contributions
of each functional group present within the drug structure. The primary purpose of
this section is to identify those functional groups that confer water solubility and those
that confer lipid solubility. Similar to electronic effects, the overall solubility contribu-
tion of a specific functional group can vary depending upon adjacent groups. Further
explanations with respect to the importance of water and lipid solubility, partition
coefficients, the ability to analyze a drug molecule and identify its water soluble and
lipid soluble components, the need for a balance between water and lipid solubility,
the advantages of increasing either water or lipid solubility, and common strategies
to alter solubility in a desired direction are discussed in detail in Chapter 5.
Water Soluble Functional Groups
Functional groups that enhance the water solubility of a drug molecule are often referred
to as hydrophilic functional groups. The two major properties that contribute to the
water solubility of a functional group are its ability to ionize and/or its ability to form
hydrogen bonds. Let us examine each of these properties separately. Acidic and basic
functional groups are capable of ionization and can become negatively or positively
charged, respectively. A permanently charged quaternary ammonium group can also
provide a positive charge; however, this functional group is only seen in a small number

Chapter 2: Functional Group Characteristics and Roles 15
of drug molecules. Functional group ionization imparts an increase in the water solubility
of a drug molecule. As such, it is important that you are able to identify acidic and basic
functional groups. The most common acidic and basic functional groups are shown in
Figures 2-8 and 2-9, respectively. Chapter 3 provides a more detailed explanation of the
acidic and basic nature of these groups, as well as several additional functional groups.
Sulfonamide Sulfon
y
lurea Phenol
Sulfonic Acid Phos
p
honic Acid
Carbox
y
lic Acid E-Dicarbonyl Imide
N
H
R
1
O
R
2
OO
H
O
R
2
R
1
O
H
O
R
OHP
O
OH
ROHSR
O
O
R
O
H
N
H
C
O
N
H
R
2
SR
1
O
O
N
H
R
2
S
O
O
R
1
Figure 2-8. Common acidic functional groups (acidic hydrogen atoms have been
highlighted).
Imine H
y
drazineAmidine Guanidine
Heteroc
y
clic Nitro
g
en
(
P
y
rrole Rin
g)
Heteroc
y
clic Nitro
g
en
(Q
uinoline Rin
g)
Alic
y
clic AmineSecondar
y
Ali
p
hatic
Amine
Primar
y
Ali
p
hatic
Amine
Primar
y
Aromatic
Amine
Tertiar
y
Ali
p
hatic
Amine
Figure 2-9. Common basic functional groups.
16 Basic Concepts in Medicinal Chemistry
A hydrogen bond is a specialized type of interaction between two dipoles (i.e., a
dipole-dipole interaction), and occurs whenever a hydrogen atom is able to serve as a
bridge between two electronegative atoms. In this type of bond, the hydrogen atom
is covalently bound to one atom and non-covalently bound to the other. A general
representation of a hydrogen bond is shown below. The atom that is covalently bound
to the hydrogen atom is known as the hydrogen bond donor, and the atom that is non-
covalently bound to the hydrogen atom is known as the hydrogen bond acceptor. Thus,
in the representation above, atom X is the hydrogen bond donor and Y is the hydrogen
bond acceptor. In general, oxygen and nitrogen are the most common hydrogen bond
donors and acceptors. A water molecule can act as both a hydrogen bond donor and an
acceptor as shown below. Thus, functional groups that are capable of forming hydrogen
bonds can form these types of bonds with water and increase water solubility. Functional
groups capable of forming hydrogen bonds are shown in Figure 2-10.
As will be discussed further in Chapter 6, hydrogen bonds are also very important
for the interactions between a drug and its biological target.
Lipid Soluble Functional Groups
Functional groups that enhance the lipid solubility of a drug molecule are often re-
ferred to as hydrophobic or lipophilic functional groups. Functional groups that lack
the ability to either ionize or form hydrogen bonds tend to impart a measure of lipid
solubility to a drug molecule. Common lipid soluble functional groups are shown in
Figure 2-11 and include unsubstituted aromatic rings, alkyl groups (aka aliphatic
side chains), unsaturated carbon rings (aka alicyclic rings), and halogens.
Fluorine is not included in the list of halogens because its effects on solubility
can vary. As shown in Figure 2-10, fluorine can act as a hydrogen bond acceptor and
enhance water solubility; however, the substitution of a hydrogen atom with a fluo-
rine atom often slightly enhances the lipid solubility of a drug molecule. Esters and
ethers can be viewed as lipid soluble functional groups depending upon the atoms or
groups attached to them. The easiest way to explain this is by way of an example. Let
H
y
dro
g
en bond
H
y
dro
g
en
bond
donor
H
y
dro
g
en
bond
acce
p
tor
G
G
G
O
H
H
O
H
H
O
H
H
O
H
H
Cl
N
CH
3
O
O
H
N

Chapter 2: Functional Group Characteristics and Roles 17
Figure 2-10. Functional groups capable of hydrogen bonds.
Unionized
carbox
y
lic ac id
Fluorine
Hydrogen Bond Acceptors
Carbamate
Disubstituted
carbamates
Urea
Disubstituted
Amides
Ketone EtherEster Heterocyclic nitrogens
(Pyridine ring shown)
Hydrogen Bond Acceptors and Donors
Hydrogen Bond Donors
Heterocyclic nitrogens
(Pyrrole ring shown)
Thiol
Hydroxyl Phenol Amide Primary and secondary
unionized amines
N
H
N
O
N
H
N
H
O
OH
O
N
H
O
O
O
N
N
O
F
H
N
H
O
N
H
O
O
H
H
S
O
O
O
O
R
1
R
1
R
1
R
2
R
1
R
1
R
2
R
3
R
2
R
1
R
1
R
2
R
3
R
1
R
1
R
2
R
2
R
1
R
1
R
1
R
1
R
1
R
2
R
1
R
2
R
1
R
2
us consider the three esters shown below with the assumption that the “R” group is
identical for all of the compounds.
Com
p
ound A Com
p
ound CCom
p
ound B

18 Basic Concepts in Medicinal Chemistry
The phosphate ester seen in compound A is clearly water soluble as it contains an
ionized functional group. In contrast, compounds B and C contain methyl and dode-
cenoyl esters, respectively. The oxygen atoms present in these latter two compounds
are still capable of forming hydrogen bonds and contributing to the overall water
solubility; however, the large alkyl chain in compound C makes the ester, as a whole, a
lipid soluble functional group. Compound B contains the smallest possible alkyl ester
and lies somewhere in the middle. In general, when viewing an ester and evaluating
its overall water/lipid solubility, you need to consider both the CO
2
portion as well as
what is connected to it. The same is true for ether functional groups.
Steric Effects
Each functional group has a finite size or steric dimension that contributes to the overall
conformation or three-dimensional shape of a given drug molecule. Obviously, some
functional groups are larger and more bulky than others, and it is impossible for two
atoms or functional groups to occupy the same space. Additionally the size and shape
of each functional group must be able to be accommodated for by the binding sites
n
Alkenes
Alk
y
nes
Alk
y
l chains
(
Ali
p
hatic chains
)
Aromatic rin
g
s
and rin
g
s
y
stems
Alic
y
lic rin
g
s
(
C
y
clo
p
ent
y
l and
C
y
clohex
y
l shown
)
Halo
g
ens
EtherEster
Figure 2-11. Lipid soluble functional groups.

Chapter 2: Functional Group Characteristics and Roles 19
present at its biological target. The addition of functional groups to a drug molecule
based primarily upon their steric effects can provide a number of therapeutic benefits
for a drug molecule including:
• increased selectivity for its biological target
• enhanced binding interactions with its biological target
• favorable alteration of its rate of metabolism
Increased Selectivity
We have already seen one example of this with acetylcholine and bethanechol (Figure
2-1). While acetylcholine non-selectively interacts with both muscarinic and nicotinic
receptors, the addition of a methyl group to the beta carbon of acetylcholine provides
bethanechol, an analog that is selective for muscarinic receptors. The selectivity results
from the fact that the muscarinic receptor has additional space that can accommodate
an extra methyl group at this position, whereas the nicotinic receptor does not.
An additional example can be seen with -adrenergic agonists and antagonists.
In addition to a number of other physiological effects, agonist activity at
1
recep-
tors produces contraction of vascular and cardiac smooth, whereas agonist action at
2
receptors produces bronchial smooth muscle relaxation. Therefore, antagonists at
the
1
receptor are useful in the treatment of hypertension and other cardiovascular
disorders, while agonists at the
2
receptor are useful in the treatment of asthma
and chronic obstructive pulmonary disease (COPD). Non-selective antagonists
can cause unwanted adverse drug reactions due to their ability to constrict bronchial
smooth muscle and exacerbate asthma and COPD. On the other hand, non-selective
agonists can cause unwanted adverse drug reactions due to their ability to constrict
vascular and cardiac smooth muscle. Similar to the example with acetylcholine and
bethanechol, selectivity between
1
and
2
receptors can be achieved by the presence
or absence of a methyl group. Shown in Figure 2-12 are the structures of atenolol, a
selective
1
receptor antagonist, and albuterol, a selective
2
receptor agonist.
Iso
p
ro
py
l
Grou
p
t-But
yl
Grou
p
Albuterol
Atenolol
Figure 2-12. Atenolol and albuterol.

20 Basic Concepts in Medicinal Chemistry
While there are a number of structural features responsible for this selectivity,
let us focus on the size of the alkyl group on the secondary amine. The presence of
the additional methyl group seen in the t-butyl group of albuterol is very important
for selective interaction with the
2
receptor. In contrast, the absence of this methyl
group in the isopropyl group of atenolol allows it to be more selective for
1
receptors.
Enhanced Binding Interactions
The interaction between a drug molecule and its biological target relies upon the
ability of the drug molecule to adopt a conformation that is complimentary to the
three-dimensional shape of the biological target. Some drugs have a considerable
amount of flexibility and can orient their functional groups in a variety of different
conformations. The energy released when a drug molecule begins to interact with
its biological target can be used to allow it to rotate about its single bonds to adopt
the specific conformation required by the biological target. However, if the energy
required to adopt the proper conformation is similar to or exceeds the energy released
through the binding interaction, then the overall binding affinity of the drug for its
biological target can be significantly diminished. This can lead to a decrease in the
overall potency of the drug and the need to administer higher doses.
One way to enhance the interaction of a drug with its biological target is to decrease
the conformational flexibility and essentially lock the drug in its active conformation.
This concept will be discussed in more detail in Chapter 7; however, one way to ac-
complish this is by adding adjacent functional groups that sterically hinder the rota-
tion of specific bonds. An example of this can be seen in the comparison of diclofenac
and fenoprofen, two non-steroidal anti-inflammatory drugs (NSAIDs). Both of these
drugs exert their mechanism of action by inhibiting the enzyme cyclooxygenase. As
illustrated in Figure 2-13, both drugs interact with cyclooxygenase through three
key interactions.
Lower phenyl ring
Lack of ortho groups
allows free rotation
of highlighted bonds
Ionized
carboxylic acid
Diclofenac
Top, adjacent
aromatic ring
Ortho groups
lock perpendic ular
conformation
Ionized
carboxylic acid
Top, adjacent
aromatic ring
Lower phenyl ring
Feno
p
rofen
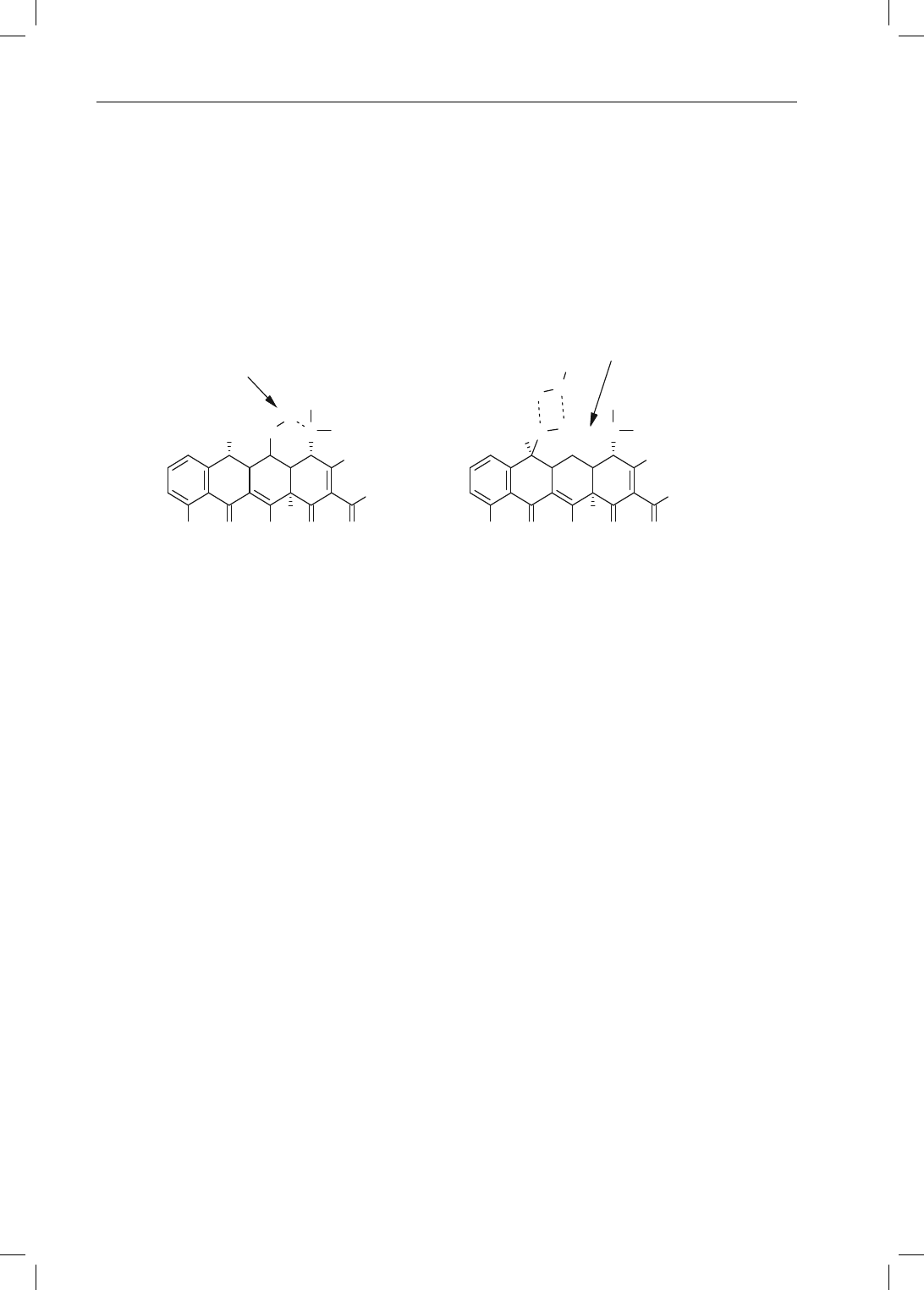
Figure 2-13. A comparison of diclofenac and fenoprofen interactions with cyclooxygenase.

Chapter 2: Functional Group Characteristics and Roles 21
The negatively charged carboxylic acid forms an ionic bond with a positively
charged cation within the enzyme. The top or adjacent aromatic ring interacts with
a hydrophobic pocket within the enzyme, and the lower phenyl ring interacts with
a second hydrophobic pocket within the enzyme. A key component of this binding
interaction is that the lower phenyl ring binds perpendicular to the top aromatic ring.
The key difference between these two drugs is that the structure of diclofenac contains
two ortho chloro groups. These two functional groups sterically lock diclofenac in its
required active conformation. In comparison, the lower ring of fenoprofen is unsub-
stituted which allows for free rotation about the indicated bonds. While fenoprofen
can still interact with cyclooxygenase, it requires energy to adopt the required active
conformation. As such, its affinity for cyclooxygenase is less than that observed for
diclofenac. From a therapeutic perspective, this difference can be seen in the normal
doses used for these two agents. The normal dose of diclofenac is 50 mg BID or TID,
while the normal dose of fenoprofen is 400 to 600 mg TID or QID. While there are
a number of factors that contribute to the dosing of these to drugs, the steric effect
seen in diclofenac definitely plays a key role.
Alteration of Metabolism
Similar to the interaction between a drug and its biological target, the metabolism
of a drug molecule requires it to interact with the active site of the enzyme that cata-
lyzes its metabolism. Steric hindrance is a common strategy used to block or slow a
specific metabolic pathway. In this approach, additional atoms are added adjacent to
the functional group undergoing metabolism in order to block the interaction of the
drug molecule with the enzyme carrying out the metabolic transformation. In many
cases, these additional atoms need not be very large. Returning back to our example
of acetylcholine and bethanechol (Figure 2-1), the additional methyl group seen on
bethanechol prevents the enzyme acetylcholinesterase from cleaving the ester bond.
Acetylcholine: Ester hydrolysis
oc c urs very rapidly
Bethanechol: Highlighted adjacent
methyl group blocks hydrolysis
CH
3
CH
3
CH
3
CH
3
N
+
CH
3
O
OO
CH
3
CH
3
CH
3
N
+
OCH
3
Another example of this concept can be seen with the cephalosporin class of
antibacterial agents (Figure 2-14). This class of drug molecules requires an intact
-lactam ring in order to exert their antibacterial action. Some bacteria can produce
-lactamase, an enzyme that catalyzes the hydrolysis of this ring and thereby inacti-
vates the cephalosporin. Cephalosporins, such as cephalexin, that can be inactivated
in this manner are known as -lactamase sensitive. This inactivation can be blocked

22 Basic Concepts in Medicinal Chemistry
by placing functional groups adjacent to the -lactam bond. As seen in Figure 2-14,
the methoxy group of cefoxitin produces steric hindrance and permits the drug to be
resistant to the actions of -lactamase.
E-Lactamase
Active cepalosporin with
int ac t E-lactam ring
Inactive cepalosporin with
hydrolyzed E-lactam ring
Steric hindranc e
Cefoxitin
E-Lactamase resistant
Cephalexin
E-Lactamase sensitive
Figure 2-14. The role of steric hindrance in inhibiting -lactam destruction by -lactamase.
FINAL CONSIDERATIONS FOR FUNCTIONAL GROUPS
ON DRUG MOLECULES
There are three key points that need to be emphasized with regard to the concepts and
examples discussed previously. First, the same functional group can provide different
effects on different drug molecules. We have already seen this in several of the prior
examples. The addition of a simple methyl group can result in a number of therapeutic
benefits depending upon the drug molecule and the location of the methyl group.
It can increase the selectivity of a drug for one biological target over another. This
was seen in earlier discussions of bethanechol compared to acetylcholine (Figure 2-1)
and atenolol compared to albuterol (Figure 2-12). Addition of a methyl group can
also increase the potency of a drug. This was previously discussed in the comparison
of lovastatin to simvastatin (Figure 2-1). An additional example can be seen in the
comparison of morphine with its N-desmethyl metabolite. The N-methyl group natu-
rally present on morphine enhances its potency approximately four fold as compared
to N-desmethylmorphine (Figure 2-15). Finally, the addition of a methyl group can
sterically block metabolism and thus increase the duration of action of a specific drug
molecule. This was described previously in the addition of the methyl group to ace-

Chapter 2: Functional Group Characteristics and Roles 23
tylcholine. An additional example of this concept is demonstrated when comparing
testosterone with methyltestosterone (Figure 2-15). Testosterone is a naturally occur-
ring androgenic hormone; however, it cannot be taken orally due to rapid oxidation of
the C
17
hydroxyl group to an inactive ketone. Addition of a methyl group at the C
17
position converts the secondary hydroxyl group into a tertiary hydroxyl group. This
blocks oxidative metabolism and allows methyltestosterone to be administered orally.
R = CH
3
: Mor
p
hine
R = H: Desmeth
y
lmor
p
hine
C
17
H
y
dox
y
l
g
rou
p
R = H: Testosterone
R = CH
3
: Meth
y
ltestosterone
Figure 2-15. Morphine and methyltestosterone: the advantages of an additional methyl
group.
Second, a single functional group may sometimes serve two distinct purposes.
Examples of this can be found in three of the drugs already discussed in this chapter.
The methyl group of bethanechol serves to both increase selectivity and prevent me-
tabolism. The two ortho chloro groups present on the lower phenyl ring of diclofenac
(Figure 2-13) provide steric hindrance and lock diclofenac in its active conformation.
Additionally, due to their electron withdrawing properties, these halogens deactivate
this ring from oxidative metabolism and allow it to have a longer duration of action
than fenoprofen. Finally, the primary amine of cephalexin provides acid stability of the
-lactam ring and enhances its ability to enter gram negative bacteria. In the acidic
environment of the stomach, this primary amine will be protonated and will act as
an electron withdrawing group. Similar to the ether oxygen of penicillin V (Figure
2-7), this electronic effect will prevent the acid catalyzed destruction of the -lactam
ring. Additionally, this primary amine will be primarily ionized at all physiologically
relevant pH environments. This enhances the water solubility of cephalexin and bet-
ter allows it to be transported by porins, the hydrophilic protein channels that allow
access of antibiotics to gram negative bacteria.
p
rimar
y
amine
Ce
p
halexin
24 Basic Concepts in Medicinal Chemistry
Finally, it is important to reemphasize that the properties of a functional group can
be altered by adjacent functional groups. Examples of this were presented earlier with
the discussion of electronic effects, so let us look at one final example that involves
solubility. Tetracycline and doxycycline (Figure 2-16) have the same molecular for-
mula (C
22
H
24
N
2
O
8
) and are chemically identical with the exception that tetracycline
contains a C
6
hydroxyl group, whereas doxycycline contains a C
5
hydroxyl group.
56
Internal hydrogen
bond
56
Dist anc e t oo far
to form internal
hydrogen bond
Doxycycline
Tetrac
y
cline
OH
OH
OH
CH
3
H
H
O
H
O
OO
NH
2
O
OH
N
OH
O
OH
NH
2
O
OH
OH
CH
3
O
O
H
N
CH
3
CH
3
CH
3
CH
3
Figure 2-16. Tetracycline and doxycycline.
Despite their structural similarities, doxycycline is much less water soluble than
tetracycline. As shown in Figure 2-16, the C
5
hydroxyl group of doxycycline is able to
form an internal hydrogen bond with the adjacent tertiary amine. This decreases the
ability of this hydroxyl group to form hydrogen bonds with water and also decreases
the ability of the amine to ionize since its lone pair of electrons is involved in this
internal hydrogen bond. In tetracycline, the hydroxyl group is no longer adjacent to
the tertiary amine and is therefore not able to form hydrogen bonds with it. As such,
it is much more available to form hydrogen bonds with water, and the lone pair of
electrons of the tertiary amine is much more available to interact with a proton to
become ionized. Thus, the position of this hydroxyl group, as well as the presence
or absence of an adjacent functional group, plays an important role in its chemical
properties. From a therapeutic perspective this minor change in the position of a single
functional group results in a number of benefits. The decrease in water solubility seen
in doxycycline allows it to have better oral absorption, enhanced penetration into
bacteria, and a longer duration of action.
A REVIEW OF FUNCTIONAL GROUPS PRESENT ON
AMINO ACIDS
As initially mentioned in Chapter 1, amino acids serve as the building blocks for a
small number of peptide based drug molecules. Amino acids also serve a much larger

Chapter 2: Functional Group Characteristics and Roles 25
role as the building blocks for proteins and enzymes. Among a plethora of other func-
tions, proteins and enzymes serve as biological targets for drug molecules. As such, it
is important to review the functional groups present on the twenty naturally occurring
amino acids, as well as the functional groups present on commonly modified amino
acids. A more in-depth discussion of drug binding interactions between drug mol-
ecules and their biological targets is provided in Chapter 6. The primary goal here is
to identify the key chemical properties of the functional groups present in amino acids
Summary of Key Points to Consider When Evaluating Functional
Groups
1. Every atom within the structure of a drug molecule is part of a
specifi c functional group.
2. The importance of a given functional group will vary among drug
molecules and drug classes.
a. A specifi c functional group can produce different effects on
different drug molecules.
b. It is possible for a single functional group to serve more than
one distinct purpose on a single drug molecule.
3. Each functional group has an electronic effect, a solubility effect,
and a steric effect.
a. It is impossible for a functional group to alter only one of
these properties without affecting the others.
b. The relative importance of these three properties will vary
depending upon the functional group.
c. The overall electronic effect of a given functional group
depends on both its ability to participate in resonance
delocalization and its intrinsic inductive effect.
d. Some electron donating groups can also act as nucleophiles,
whereas some electron withdrawing groups can also act as
electrophiles.
e. The two key properties that contribute to the water solubility
of a functional group are its ability to ionize and its ability to
participate in hydrogen bonding interactions.
f. Each functional group has a fi nite size or steric dimension that
contributes to the overall conformation of a given drug molecule.
4. The overall effect of a given functional group is dependent upon
other adjacent or surrounding functional groups.
5. Functional groups can be altered to provide specifi c therapeutic
benefi ts.
26 Basic Concepts in Medicinal Chemistry
and therefore in proteins. Similar to functional groups present on a drug molecule,
the functional groups present on amino acids have electronic, solubility, and steric
effects. The primary difference is that unlike drug molecules, the functional groups
present on the amino acids of a protein or enzyme can’t be changed. In many of the
examples discussed in this chapter, we examined the advantages that could be gained
by adding, removing, or altering a functional group. Since these types of alterations
do not occur with the amino acids present in proteins and enzymes, this section has
been intentionally placed at the end of the chapter.
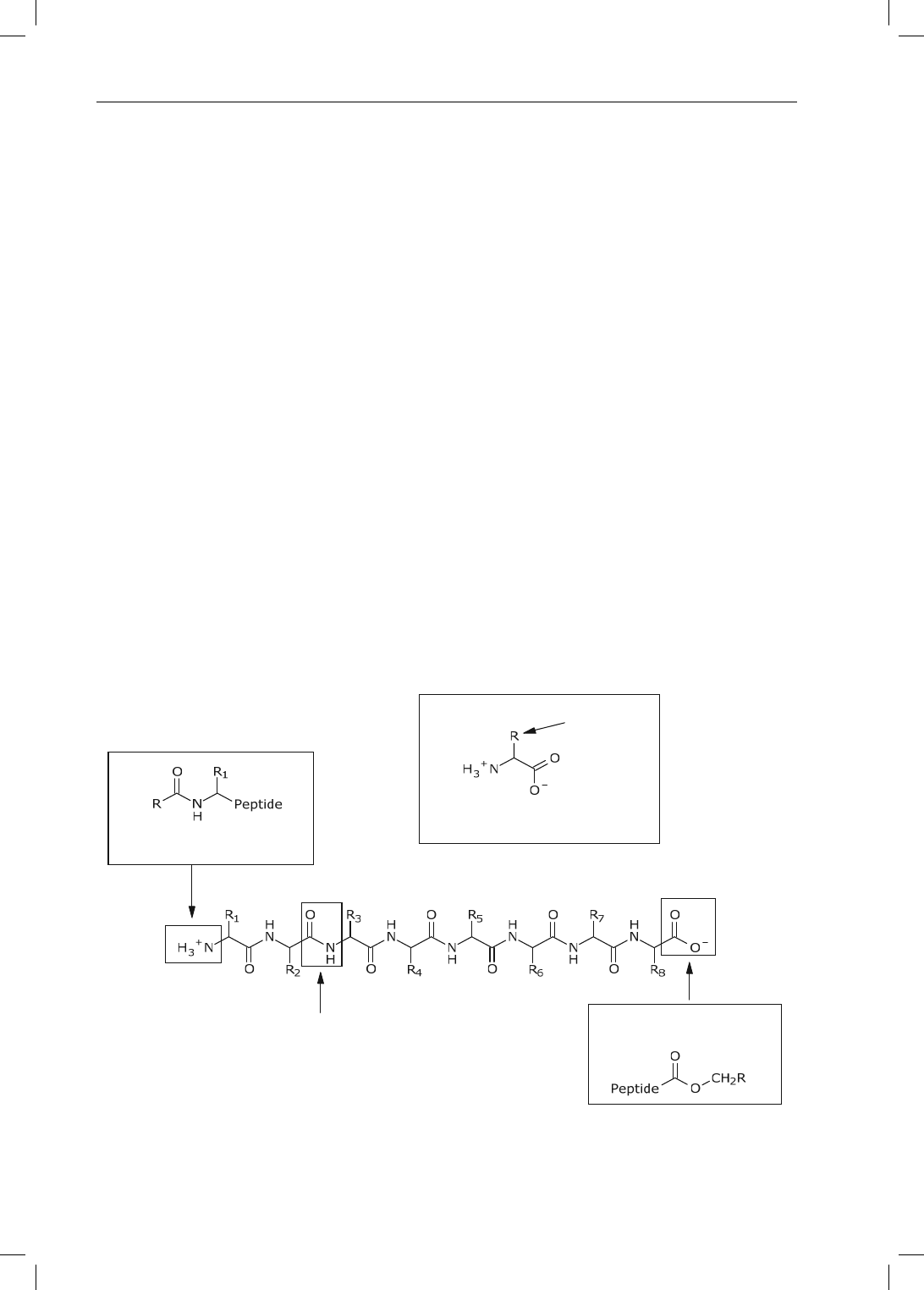
As shown in Figure 2-17, each amino acid contains a primary amine, a carbox-
ylic acid, and a side chain at the -carbon. While this section focuses primarily on
the functional groups present on the side chains, let us briefly examine the amines
and carboxylic acids of amino acids. These functional groups are used to form amide
bonds, or peptide bonds. A protein therefore consists of one or more peptide chains
that contain numerous amide bonds, a primary amine at one end, and a carboxylic
acid at the other end. These ionizable groups are often masked as amides and esters
in order to avoid degradation by endogenous exopeptidases. The multiple amides that
comprise the peptide backbone are polar functional groups and are extremely impor-
tant for forming internal hydrogen bonds. Once the three-dimensional conformation
of the peptide has been established, the functional groups present on the side chains
are able to interact with drug molecules or other endogenous moieties.
Amide
(Seven t otal in peptide c hain)
Terminal carboxylic acid
(Often converted to ester)
Terminal amine
(Often converted to amide)
General Structure
of Amino Acid
D
Side chain
Figure 2-17. General structures of amino acids and peptide chains.

Chapter 2: Functional Group Characteristics and Roles 27
Glycine is the smallest and simplest of all of the amino acids. Its side chain is a
hydrogen atom; although some texts view glycine has having no side chain. It is es-
sentially neutral in terms of electronic or solubility effects. From a steric perspective,
it really has no steric hindrance and conveys flexibility to a peptide chain. Please note
that for glycine and all of the other amino acids to follow, the primary amine and the
carboxylic acid are intentionally drawn in their unionized forms since they are normally
involved in the formation of a peptide bond.
Gl
y
cine
Alanine, valine, leucine, and isoleucine all contain aliphatic, lipid soluble, hydrocarbon
side chains and have varying steric bulk. The side chains on these amino acids are most
important for forming lipid soluble pockets within a protein or enzyme and interacting
with other hydrocarbon chains on drug molecules or endogenous compounds. Addi-
tionally, the varying steric bulk on these amino acids can either block or allow access to
certain portions of a protein depending upon which amino acid is present.
Alanine Valine IsoleucineLeucine
Proline is unique from all of the other naturally occurring amino acids in that it
contains a secondary amine. Similar to the previous four amino acids, proline has a
lipid soluble, hydrocarbon side chain. The key difference is that the side chain forms
an alicyclic ring with the nitrogen atom. The most important feature of proline is that
the alicyclic ring produces a significant steric affect that causes protein chains to kink
or bend. In addition, due to the alicyclic ring, natural human proteases cannot cleave
peptide bonds that include a proline amino acid.
Proline
N
H
CO
2
H
Phenylalanine and tryptophan contain aromatic rings and similar to the aliphatic
and alicyclic amino acids are lipid soluble. Due to the aromaticity of both rings, these
functional groups have a greater electronic nature and can interact with aromatic rings
present on drug molecules. A key difference between these two amino acids is the

28 Basic Concepts in Medicinal Chemistry
presence of the nitrogen atom in the bicyclic indole ring of tryptophan. This nitro-
gen is not basic due to the fact that its lone pair of non-bonding electrons is required
for the aromaticity of the bicyclic ring. This donation of electrons causes the indole
ring of tryptophan to be more electron rich than the phenyl ring of phenylalanine.
Additionally, the nitrogen atom in the indole ring can act as a hydrogen bond donor
and therefore can participate in hydrogen bonds. The phenyl ring of phenylalanine
cannot do this. From a steric perspective, both rings are larger than the side chains of
the above aliphatic or alicyclic amino acids.
Phen
y
lalanine
Tr
yp
to
p
han
Serine, threonine, and tyrosine all contain hydroxyl groups within their side chains.
These functional groups are very important due to their ability to form hydrogen
bonds as both hydrogen bond donors and acceptors with water, as well as with other
functional groups present within drug molecules. Both serine and threonine are classi-
fied as water soluble uncharged amino acids. Tyrosine is often classified as an aromatic
amino acid along with phenylalanine and tryptophan; however, it is included here
due to its phenolic hydroxyl group. It is more water soluble than the phenyl ring of
phenylalanine. Additionally, the hydroxyl groups of all three of these amino acids are
nucleophiles and are important for specific mechanisms in biochemical pathways and
for the mechanisms of action of some drug molecules. Due to their nucleophilic nature,
these functional groups are often phosphorylated as part of a regulatory mechanism
or are added to increase the number of negative charges associated with the protein
or enzyme. An example of this is seen with phosphoserine.
Threonine
Serine Phos
p
hoserineT
y
rosine
Cysteine and homocysteine are very similar to serine. Cysteine is one of the twenty
naturally occurring amino acids, whereas homocysteine is a precursor to methionine
and is involved in several biochemical pathways. Both of these are water soluble un-

Chapter 2: Functional Group Characteristics and Roles 29
charged amino acids; however, their ability to form hydrogen bonds is weaker than
that observed with serine, threonine, and tyrosine. They are also very nucleophilic.
The ability of cysteine to form disulfide bonds is essential for the three-dimensional
conformation of a number of peptides and proteins. Methionine, which contains a
thioether in its side chain, is much less reactive and is often grouped along with ala-
nine, valine, leucine, and isoleucine as a lipid soluble amino acid. It is included here
because of its relationship to cysteine and homocysteine.
Disulfide bond
between two
c
y
steines
Methionine Homoc
y
steineC
y
steine
As the names imply, aspartic acid and glutamic acid are the two naturally oc-
curring acidic amino acids. These carboxylic acids will be primarily ionized in most
physiological environments, with the stomach being the primary exception. These
functional groups are water soluble and provide negative charges for ionic interactions
with positively charged functional groups or metal ions. The only difference between
these two amino acids is the additional methylene carbon present in glutamic acid.
Thus from a steric perspective, glutamic acid has somewhat more steric bulk than
does aspartic acid. Similar to the terminal carboxylic acid shown in Figure 2-17, these
amino acids can be esterified to remove their ability to be ionized and decrease their
overall water solubility.
Glutamic Acid
As
p
artic Acid
Asparagine and glutamine are the amide analogs of aspartic acid and glutamic
acid, respectively. These amino acids are similar to serine, cysteine, and threonine in
that their side chains are water soluble and not able to be ionized. The amide groups
can serve as hydrogen bond donors and acceptors. Since the lone pair of non-bonding
electrons on the amide nitrogen are involved in resonance with carbonyl group, they
are not very nucleophilic.
30 Basic Concepts in Medicinal Chemistry
GlutamineAs
p
ara
g
ine
Finally, lysine, arginine, and histidine are the three naturally occurring basic amino
acids. The relative basicities of these functional groups is discussed in more detail in the
next chapter; however, both lysine and arginine are primarily ionized in all physiological
environments and can provide positive charges for ionic interactions with negatively
charged functional groups. Histidine is less basic than either lysine or arginine and is
primarily unionized in most physiologically relevant environments. Histidine can act
as either a hydrogen bond donor or a hydrogen bond acceptor. The functional groups
on all three amino acids enhance water solubility. The primary amine of lysine can be
methylated to form a quaternary amine, trimethyllysine. This provides a permanent
positive charge within a protein.
HistidineL
y
sine
Trimeth
y
ll
y
sineAr
g
inine

Chapter 2: Functional Group Characteristics and Roles 31
REVIEW QUESTIONS
1. For the structure of napsagatran, name all of the boxed functional groups in the grid
below.
Na
p
sa
g
atran
A
B
C
D
E
F
G
S
N
N
OH
O
O
N
H
N
NH
NH
2
H
H
OO
Box Functional Group Name
A
B
C
D
E
F
G
2. For the structure of clobetasol, name all of the boxed functional groups in the grid
below.
A
B
C
D
E
F
Clobetasol
O
CH
3
O
CH
3
O
Cl
O
OH
F

32 Basic Concepts in Medicinal Chemistry
Box Functional Group Name
A
B
C
D
E
F
3. The structural features found within H
2
receptor antagonists include a basic functional
group that is protonated at physiological pH (A), an aromatic ring (B) and a terminal
non-basic polar functional group (C) that is separated from the aromatic ring by the
equivalent of a four-carbon chain. The terminal non-basic polar functional group
participates in a key ion-dipole interaction with an ionized carboxylic acid found in the
binding region within the H
2
receptor.
-
C
B
A
A. Identify all of the functional groups that are present in the structure of famotidine.
Famotidine
(
Pe
p
cid
)
B. Guanidines and amidines that are substituted with electron withdrawing groups have
signifi cantly decreased basicity compared to unsubstituted guanidines (pKa ~ 12.5)
and amidines (pKa ~ 9) and are unprotonated (unionized) at physiological pH. Name
the two electron withdrawing functional groups found within famotidine.
C. The boxed functional group (show below) participates in a key ion-dipole interaction
with an ionized carboxylic acid group found in the H
2
receptor binding region.
Provide a brief rationale why this functional group is not protonated (ionized) at
physiological pH.

Chapter 2: Functional Group Characteristics and Roles 33
Famotidine
(
Pe
p
cid
)
4. JB, a 47-year-old mother of three children under the age of eight, has experienced
prolonged periods of constipation after the birth of her last child 3 years ago. She
has changed her diet to include more fi ber and has increased her fl uid intake as
recommended by her physician. There does not appear to be any correlation between
what she eats and the length of time she experiences constipation. She does stay away
from foods that are binding, including bananas, rice, and apples. Her physician suggests
that she try taking Enulose
®
, and if that doesn’t work, he may recommend glycerin
suppositories.
Lactulose
(
Enulose
)
A. Consider the structure of Enulose
®
drawn above when completing the grid below.
Name of Three Oxygen
Containing Functional
Groups
Hydrophilic and/or
Hydrophobic
Contribution to Water
Solubility and/or Lipid
Solubility
Hydrogen Bond Acceptor,
Donor, Both or Neither
B. Enulose
®
is supplied as an aqueous solution of lactulose 10 mg/15 mL and is
typically administered thre e or four times daily (30–45 mL/dose). Based on your
functional group evaluation on the previous page, provide a structural rationale for
why Enulose
®
can be formulated as an aqueous solution.
C. Mechanistically, lactulose is classifi ed as an osmotic laxative, which means that
it is able to draw water into the intestine to help soften the stool. Based on your
functional group evaluation on the previous page, provide a structural rationale for
why Enulose
®
is able to draw water into the intestine.

34 Basic Concepts in Medicinal Chemistry
D. Using the structure below, show (and label) examples of the following interactions.
a. Drug/water interaction: drug = hydrogen bond acceptor; water = hydrogen
bond donor
b. Drug/water interaction: drug = hydrogen bond donor; water = hydrogen bond
acceptor
c. Drug/water interactions: dipole/dipole interaction
Note: When appropriate, be sure to draw in the correct arrow and partial charges.
Lactulose
(
Enulose
)
5. A 24-year-old male comes into the pharmacy and asks you for a recommendation for a
treatment for the itching and burning he has recently noticed on both feet. He indicates
that he would prefer a cream rather than a spray or a powder. Your recommendation to
this patient is to use terbinafi ne (Lamisil
®
), a very effective topical antifungal agent that is
sold over the counter.
Terbinafine
(
Lamisil
)
A. Identify all of the structural features present in terbinafi ne and complete the grid
below.
Name of Functional Group
Hydrophilic and/or
Hydrophobic
Contribution to Water Solubility and/or
Lipid Solubility

Chapter 2: Functional Group Characteristics and Roles 35
B. Using the information that you provided in the grid in the previous question, provide
a structural rationale for why terbinafi ne is an agent that can be applied topically.
(Hint: Agents that are administered topically must absorb into the hydrophobic
components of the skin in order to be effective.)
6. Binding interactions with a biological target can be signifi cantly impacted by the
presence or absence of functional groups that dictate a molecule’s shape. In each of
these molecules, functional groups in the ortho position of the aromatic rings force the
two ring systems to be perpendicular to one another. Circle the functional groups that
infl uence the shape of each of these molecules.
Amlodi
p
ine
Clonidine
Ox
y
metazoline
N
N
H
CH
3
OH
(H
3
C)
3
C
CH
3
7. Specifi city for a biological target can be signifi cantly impacted by the presence or
absence of functional groups that alter the size of a molecule. Epinephrine is a non-
selective agonist at
and receptors. Determine which functional group in albuterol
causes it to have receptor selectivity and identify if the functional group represents a
change in electronic or steric factors.
E
p
ine
p
hrine
Albuterol
8. The addition of one functional group can signifi cantly alter the biological activity
that a molecule exhibits. In this case, the presence of a chloro substituent allows for
dopaminergic action. Determine whether or not this change in biological activity is a
result of a steric or electronic effect and provide a rationale for your answer.
Promazine
Chlor
p
romazine

36 Basic Concepts in Medicinal Chemistry
9. Lopinavir is an HIV protease inhibitor. Structurally, this drug is considered a
peptidomimetic, in that it resembles (at least in part), the peptide substrate for the
protease. In the presence of this agent, HIV protease is unable to cleave the gag-pol
polypeptide into its functional proteins during viral assembly and budding.
A
Lopinavir
C
B
O
CH
3
CH
3
O
HOH
H
O
CH
3
CH
3
O
N
N
N
N
A. Consider the portion of the molecule designated as “A” (You will notice that the
atoms and bonds of this portion of the molecule have been bolded for you.) Which
amino acid is this? How do you anticipate that the side chain of this amino acid will
contribute to solubility and absorption of this drug?
B. Consider the portions of the molecule designated as “B” and “C,” while the normal
peptide bond sequence has been slightly changed, the side chains are unaltered.
Which amino acid do these portions resemble? How do you anticipate that the side
chain of this amino acid interacts with the biological target?
